The Phelan-McDermid Syndrome Foundation is committed to keeping our community informed about the latest scientific and medical research, including progress in therapeutic development and clinical trials. Although PMSF will not endorse a particular pharmaceutical company or treatment, we strongly believe in creating meaningful partnerships with industry so that our community is not only well-informed but also has its voice and needs included at every stage of the research process.
PYC Therapeutics recently participated in the Phelan-McDermid Syndrome Congress in Barcelona, Spain (check out our recent blog post from Diane!). We had the opportunity to meet with them the following week to hear their presentation, which they have generously shared with our PMSF community!
Since there is no recording of the presentation, Dr. Lauren will help break down the presentation for you! She offers both an overview of the progress of PYC-002 and a quick slide-by-slide summary!
Highlights
- Reminder: PYC-002 is an RNA-based therapy, specifically an ASO (Antisense Oligonucleotide) Therapy–learn more here in our previous post
- Currently, PYC-002 is at the “pre-clinical” stage of development
- PYC-002 has been tested in patient-derived cells and has demonstrated the ability to restore SHANK3 protein to “normal” levels
- PYC-002 has most recently been tested in wild-type rats, or rats that have normal SHANK3 protein expression, for safety
- Next, PYC-002 will be moving to testing PYC-002 in SHANK3-deficient rats
- Human clinical trials for PYC-002 are still expected to begin in 2026 in the United States
- Exact date and inclusion/exclusion criteria (e.g., age of participants, type(s) of deletion/mutation) have not yet been determined
- PYC-002 will be delivered by lumbar puncture (intrathecal)
- Due to the mechanism of ASOs, repeated regular dosing will occur; however, the exact treatment frequency is still to be determined, though estimated to be every 3-6 months
Slide-by-Slide Guide
Slide 3 – Highlights of what has been done and where things are going
We summarized and expanded on above

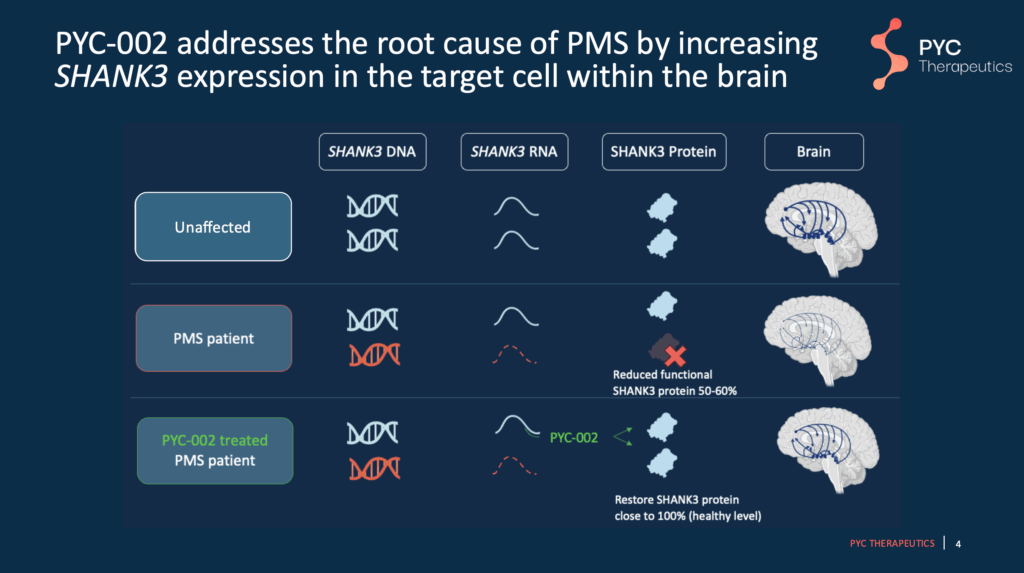
Slide 4 – Review of how PYC-002 works
PYC-002 increases SHANK3 protein in the already healthy RNA copy
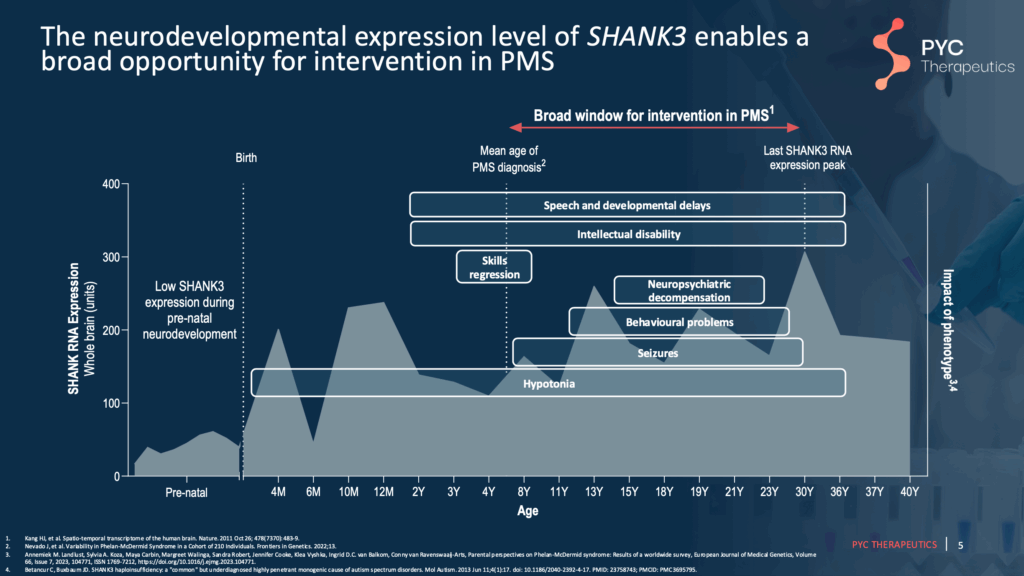
Slide 5 – Potential Optimal Treatment Timing
SHANK3 RNA expression across development (gray background) overlaid with developmental timelines of characteristic clinical features. Based on the emergence of symptoms and average age of diagnosis, there may be a broad window when PYC-002 may be most effective.
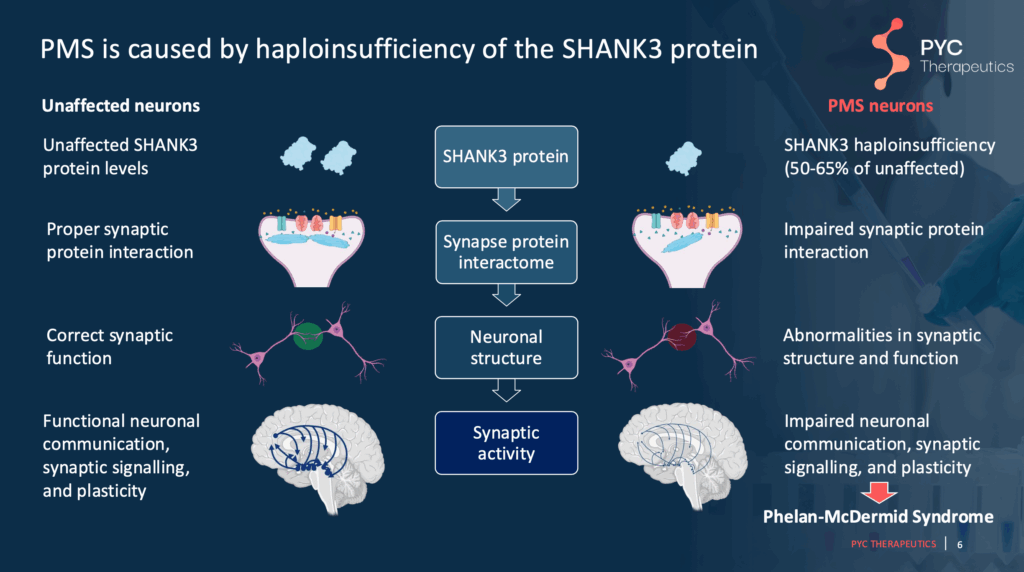
Slide 6 – Review of biological mechanisms underlying SHANK3-related Phelan-McDermid syndrome
One copy of SHANK3 gene is impacted in Phelan-McDermid syndrome, and thus there is only one functional copy of the SHANK3 (this is called “haploinsufficiency”). As a result, roughly half of the expected SHANK3 protein is expressed, leading to a cascade of effects in the brain. One of the most prominent negative effects is how brain cells, or “neurons”, form connections with each other and talk to each other via specific chemicals/molecules called neurotransmitters. Ultimately, this has a widespread impact on the brain, leading to the clinical features of Phelan-McDermid syndrome, including intellectual disability, hypotonia, etc.
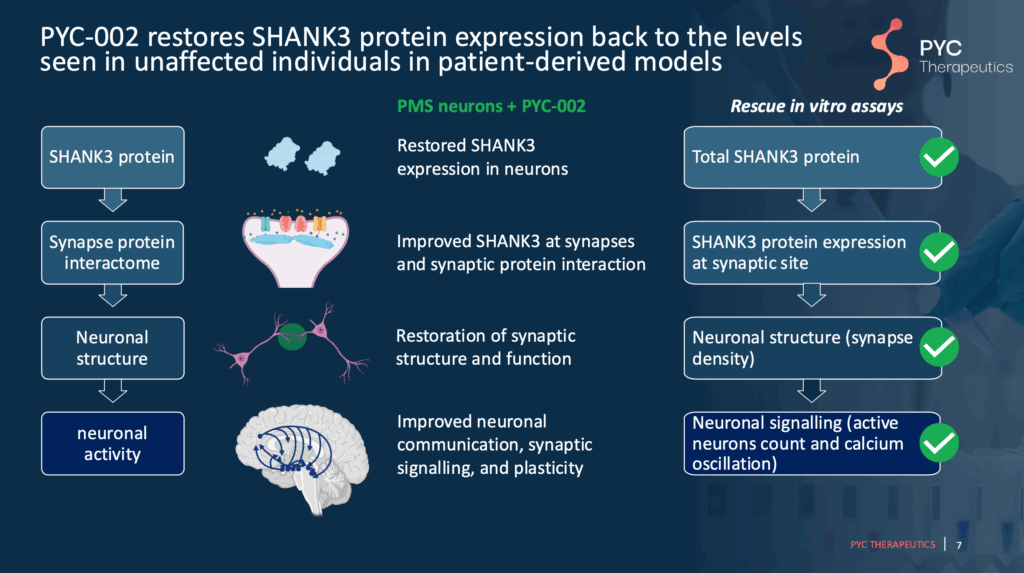
Slide 7 – How PYC-002 Works
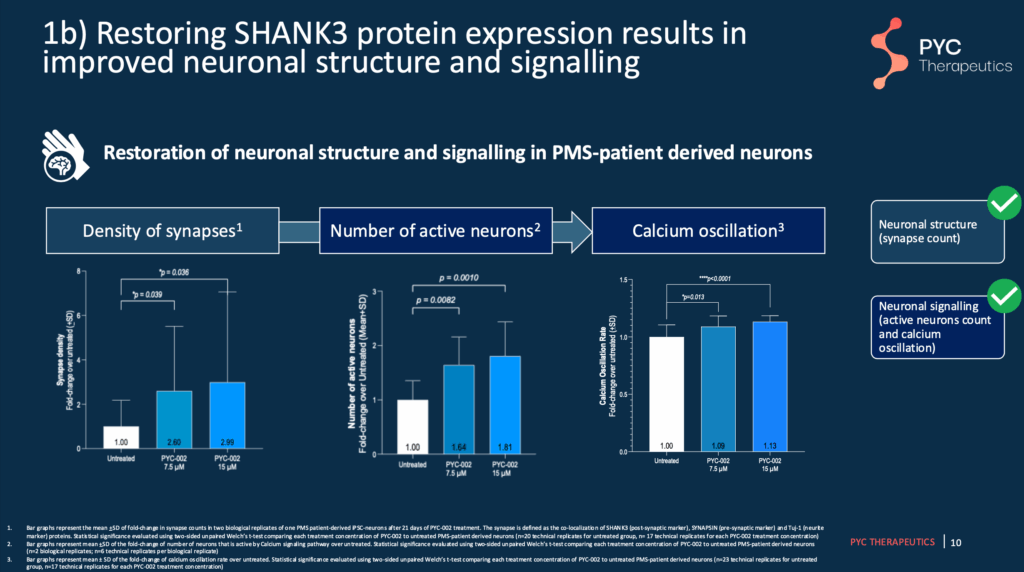
Briefly, PYC-002 increases SHANK3 protein expression in the healthy copy of the SHANK3 gene. In their previous studies, PYC-002 has been shown to increase total SHANK3 protein expression to expected “healthy” levels and improve the structure and function of the connections between neurons, or “synapses”.
Slide 8 – Efficacy and Safety Profile
The pre-clinical studies completed in patient-derived neurons as well as in unaffected rodents show that PYC-002 is safe and impacts SHANK3 gene and protein expression as desired
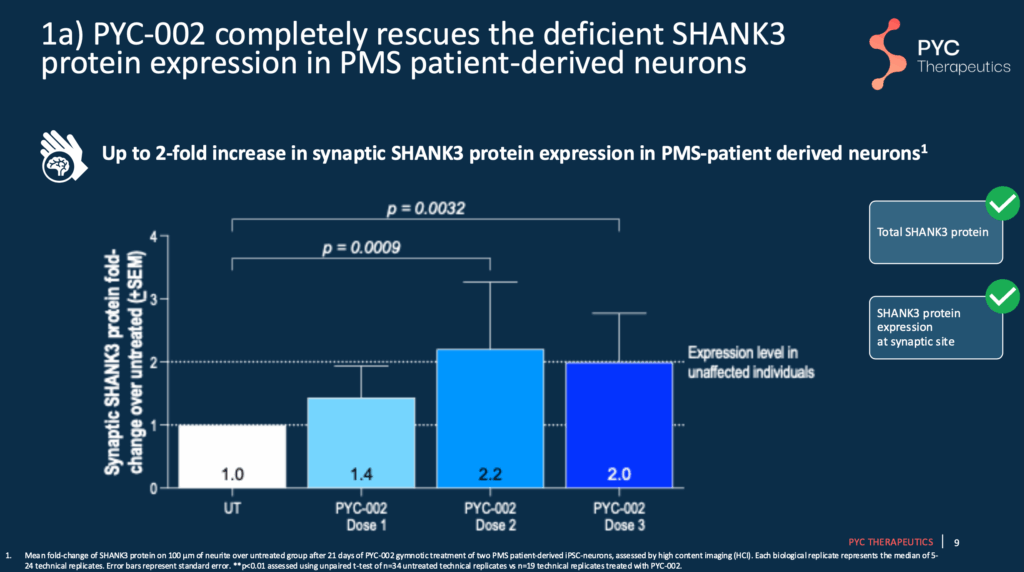
Slide 9 – 11 – Findings Using Patient-Derived Neurons
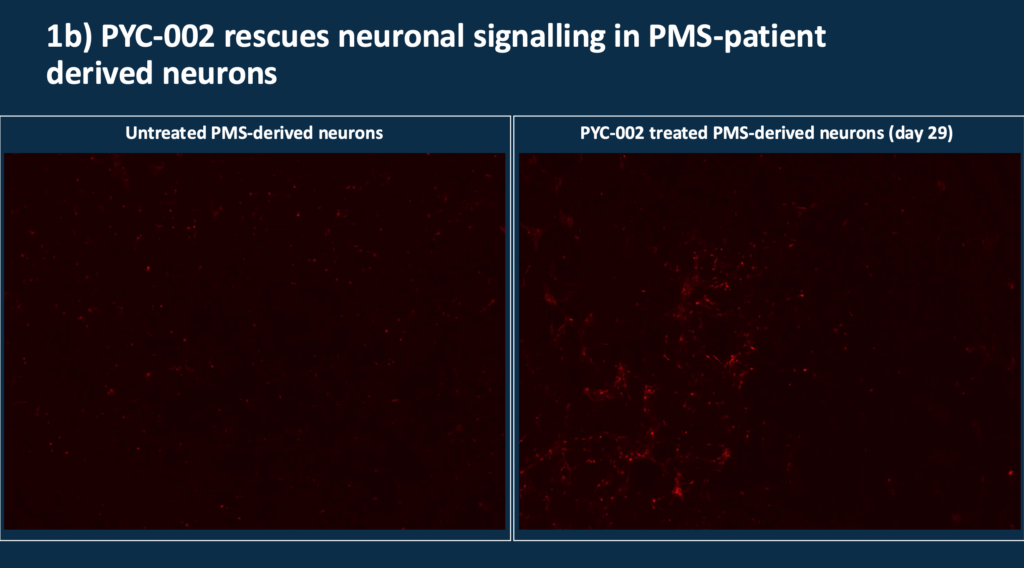
PYC-002 increases SHANK3 protein two-fold, helping increase synaptic activity and function. This is represented by the brighter red areas on the right side of Slide 11.
Slide 12 – Brain Areas Impacted in Phelan-McDermid Syndrome
Multiple brain areas have been found to be disrupted in Phelan-McDermid syndrome. These different brain areas have different primary functions.
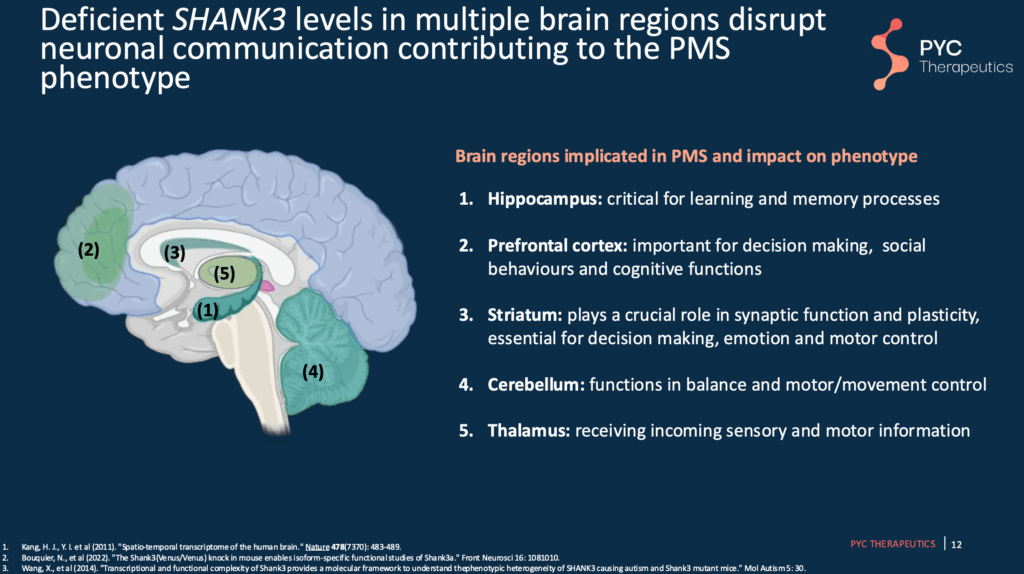
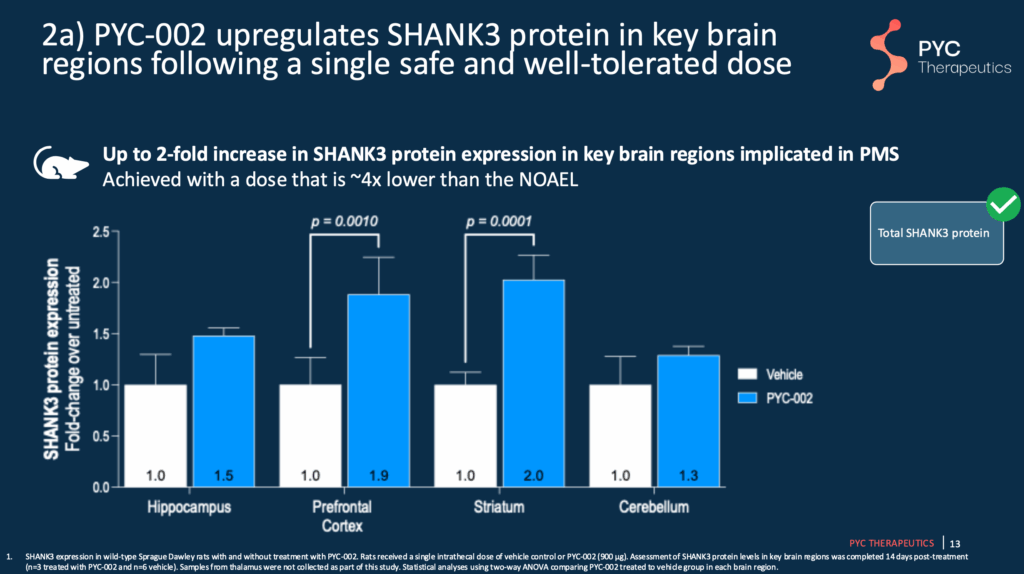
Slide 13 – PYC-002 Increases SHANK3 protein in specific brain regions
In unaffected mice (e.g., mice without SHANK3 mutation), PYC-002 increases SHANK3 protein in some of the brain areas known to be impacted in Phelan-McDermid syndrome.
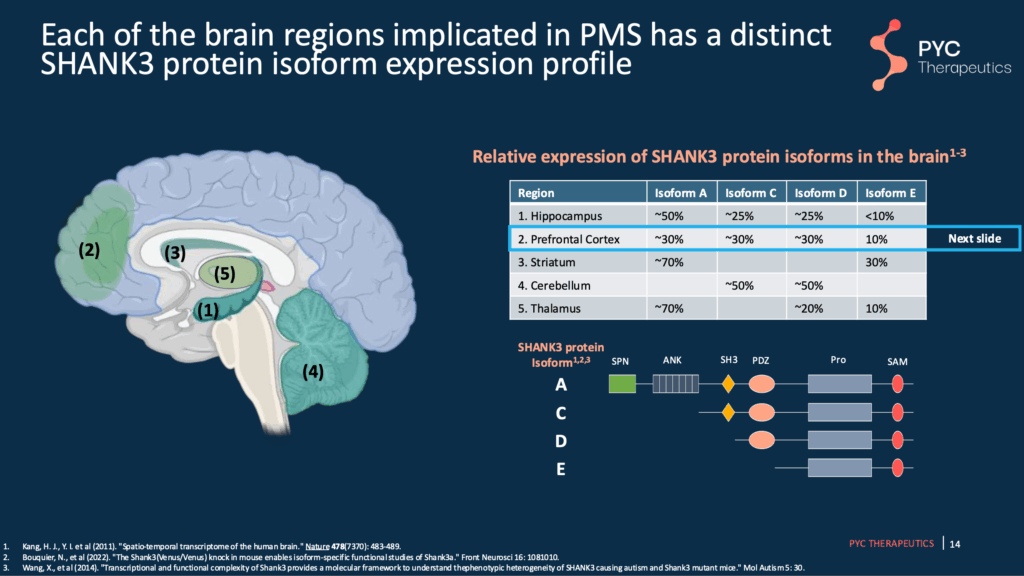
Slide 14 – SHANK3 Protein Isoforms
SHANK3 protein has different versions or variations that can have slightly different functions. These different variations are found in quantities in different parts of the brain, as shown.
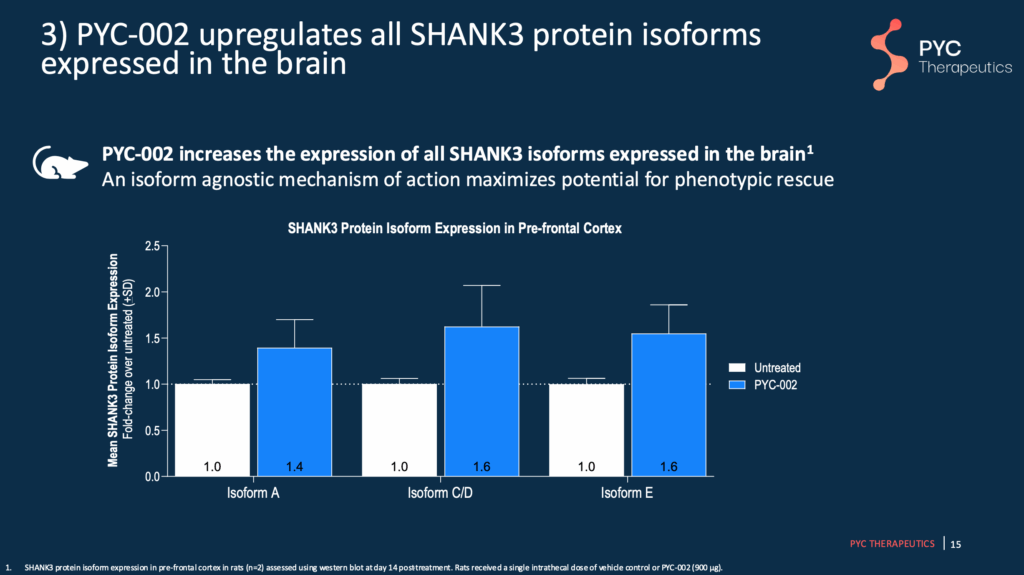
Slide 15 – PYC-002 Increases All SHANK3 protein isoforms
Using unaffected rats, PYC-002 is shown to increase all SHANK3 isoforms, thus allowing for the most coverage and potential in restoring function
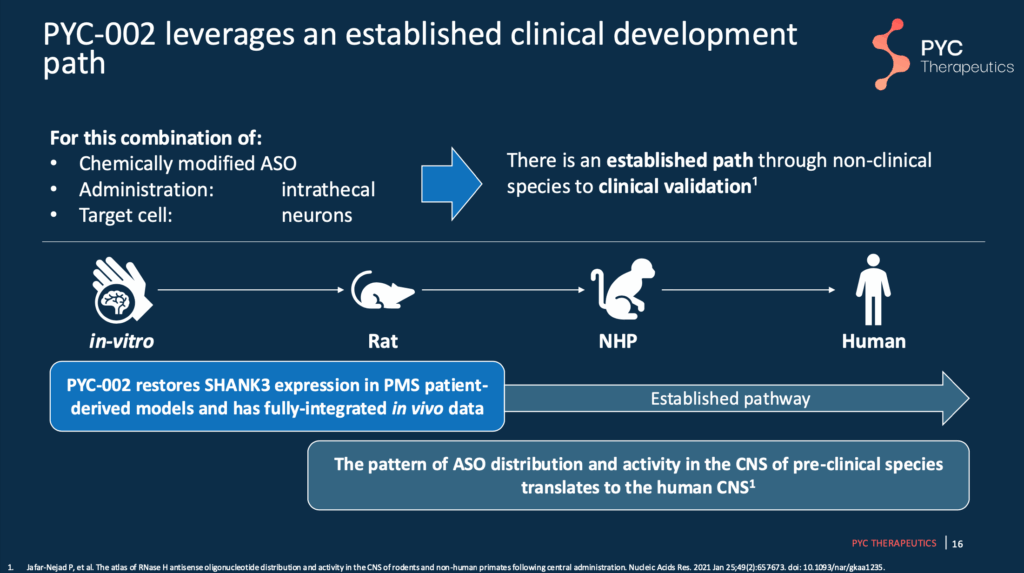
Slide 16 – PYC-002 Pipeline
Studies have been completed and shown promising findings in patient-derived neurons (“in vitro”) and unaffected wild-type rats. The next stages are to move into SHANK3-deficient rat models, unaffected non-human primates, and finally, humans.
Slide 17 – Expected PYC-002 Timeline
Additional pre-clinical studies will be completed in mid-2026. Following regulatory submission to the FDA, the first in-human trial with PYC-002 will hopefully begin by the end of 2026! Details regarding inclusion/exclusion criteria and study sites are not yet known.

