By Lauren Schmitt, PhD
January 12, 2026
With Neuren’s Phase 3 clinical trial for NNZ-2591 now open for enrollment, the recent publication of its Phase 2 trial results in Neurology: Genetics marks an important milestone in the search for a treatment for Phelan-McDermid syndrome (PMS).
Dr. Ann Neumeyer (first author on the publication) shared that, “The results from the NNZ-2591 Phase 2 study are encouraging, showing improvements on both broader neurodevelopmental measures as well as on measures tailored specifically to Phelan-McDermid syndrome. This progress has led to the launch of the first-ever Phase 3 clinical trial for PMS, which is now enrolling participants.”
What Did the Study Do?
NNZ-2591 was evaluated in 18 children and adolescents aged 3–12 years (66% male) with Phelan-McDermid syndrome in a phase 2, open-label clinical trial (NCT05025241). NNZ-2591 is a novel, orally administered compound designed to mimic a naturally occurring metabolite of IGF-1 (cyclic glycine-proline) to help restore synaptic function, which is known to be disrupted due to SHANK3 haploinsufficiency.
Participants’ PMS genotypes were classified as class I deletions (which includes deletion of only SHANK3, or deletion of SHANK3 with ARSA and/or ACR and RABL2B), class II deletions (all other terminal deletions), ring 22, unbalanced translocation, interstitial deletion, SHANK3 pathogenic variant, and other. Participants received NNZ-2591 orally twice-a-day for 13 weeks.
What is a Phase 2 trial? A study that looks at whether a treatment works and continues to check its safety in a small number of participants. Phase 2 trials help determine whether a treatment is initially promising or not.
What is an open-label trial? A study where the doctors, study team, and family all know they are receiving the active drug. There is no placebo group in an open-label trial.
What Did the Study Find?
- Was it safe? (Safety) – Yes, NNZ-2591 was well-tolerated, with most side effects (or “treatment-emergent adverse events”) being mild to moderate. The most commonly reported side effects were increased psychomotor hyperactivity, decreased appetite, fever, and sleepiness. Three of the 18 participants discontinued participation in the trial for reasons that were determined to be unrelated to the study drug. No clinically significant changes in laboratory values, electrocardiograms, or other safety parameters were reported; no deaths were reported
- Did the body process the drug? (Pharmacokinetics) – Yes, NNZ-2591 was appropriately absorbed by the body based on concentration of the study drug found in the blood.
- Did it help? (Efficacy) – Yes, improvements were observed across all PMS-specific measures as well as in quality of life, challenging behaviors, GI issues, and sleep comparing baseline to end of treatment.
- Clinicians observed improvements in receptive and expressive language, cognitive and learning abilities, social interaction, motor skills, and self-care.
- Caregivers similarly reported progress in communication, behavior, and social functioning — domains that align closely with their highest priorities.
- Reductions in the clinician-reported PMS-specific measure were observed in 7 out of 18 participants, with the greatest improvements reported in sleep and behavior.
Figure 2 from Neumeyer et al., 2025, demonstrating change in clinician- (A, C) and caregivers-reported (B, D) measures.
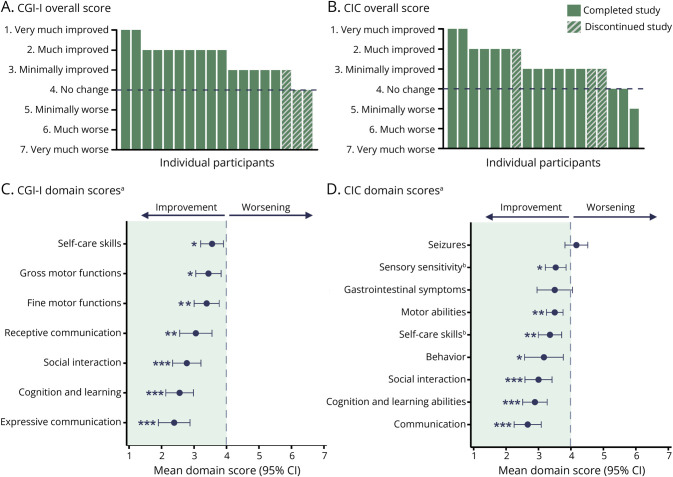
*p < 0.05; **p < 0.01; ***p < 0.001 for observed change relative to baseline vs null median of 4 (no change) based on the Wilcoxon signed-rank test. a n = 18 (unless otherwise noted). b n = 17. CGI-I and CIC overall scores and domain scores range from 1 to 7; lower scores indicate improvement (below 4), a score of 4 indicates no change, and higher scores (above 4) indicate worsening. CIC = Caregiver Impression of Change; CGI-I = Clinical Global Impression of Improvement; PMS = Phelan-McDermid syndrome.
What Does This Mean?
In an open-label trial of NNZ-2591, participants with Phelan-McDermid syndrome demonstrated improvements across several domains at the of the trial at 13 weeks. Both clinicians and caregivers reported improvements in motor skills, communication, social interaction, self-care skills and cognitive and learning. This suggests that NNZ-2591 may improve several key areas, including those identified by caregivers as being top priorities for them (communication and cognition).
It is also important to note that there is currently no gold-standard outcome assessment or syndrome-specific rating scale used in clinical trials for Phelan-McDermid syndrome. This trial developed and used five PMS-specific measures in this trial, and improvements were seen across all five of these PMS-specific measures. This suggests that these measures may be useful in evaluating efficacy in future clinical trials of NNZ-2591 as well as other treatments.
Like many Phase 2 studies in rare genetic disorders, this trial had limitations including a small sample size (n=15 completed), short duration (13 weeks), and an open-label design without a placebo control. These factors may introduce bias or limit the generalizability of findings. However, these results are still a critical step, especially when treatment options are limited.
So What’s Next?
As Dr. Liz Berry-Kravis notes, “We’re hopeful that this next step will confirm the improvements seen in Phase 2 and bring us closer to a meaningful treatment option for individuals with Phelan-McDermid syndrome.”
The launch of this first-ever Phase 3 trial for Phelan-McDermid syndrome is an important milestone in our community. The Phase 3 randomized, double-blinded, placebo-controlled trial of NNZ-2591 is open for enrollment. At the time of this blog post, there is only 1 active site, but additional sites will be added over the course of 2026.
Resources
Full Citation: Neumeyer AM, Srivastava S, Holder JL, Milad MA, Squires L, Jones NE, Glass L, Berry-Kravis E. NNZ-2591 in Children and Adolescents With Phelan-McDermid Syndrome: Single-Group, Open-Label, Phase 2 Trial Results. Neurol Genet. 2025 Dec 23;12(1):e200338. doi: 10.1212/NXG.0000000000200338. PMID: 41450730; PMCID: PMC12735298.
More information on Neuren’s Phase 3 study:
https://pmsf.org/document/neurens-phase-3-nnz-2591-trial/
https://pmsf.org/neurens-phase-3-nnz-2591-trial-for-phelan-mcdermid-syndrome-is-now-active/
https://clinicaltrials.gov/study/NCT07281079?cond=Phelan-McDermid%20Syndrome&rank=2
